You have selected a link to a website that exists outside of the IGALMIhcp.com website.
BioXcel Therapeutics, Inc. ("BioXcel") provides access to this site as
a service to IGALMIhcp.com visitors and is not responsible or liable for
the content and information presented within any non-BioXcel external site.
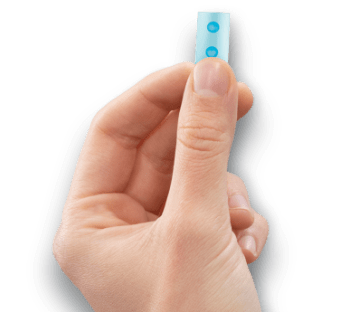
This reprint article is provided by BioXcel. It may include information
about IGALMI™ (dexmedetomidine) sublingual film that is not included
in the accompanying US Food and Drug Administration (FDA)-approved Prescribing
Information.
BioXcel does not recommend the use of IGALMI for any purposes that are
inconsistent with the Prescribing Information. Certain authors, panel members,
or contributors to this article may have acted as consultants for BioXcel.
Go to Link